Outcomes
Commitment to Quality Care
Wilmot's Blood & Marrow Transplantation Program is accredited by the Foundation for the Accreditation of Cellular Therapy and recognized for its high level of care and detailed quality management processes.
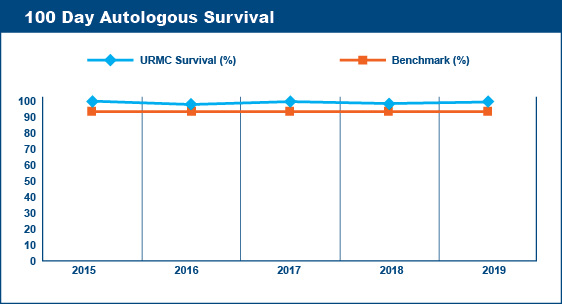
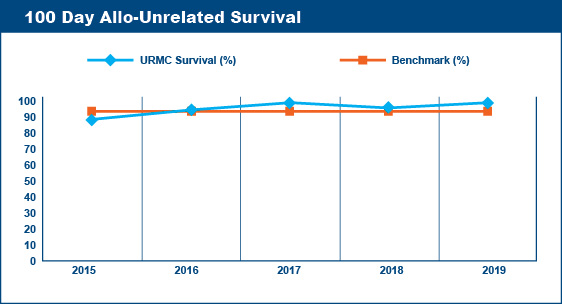
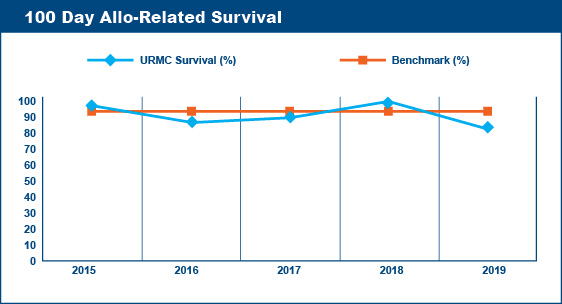
Transplant-Related (Non-Relapse) Mortality Rates
Survival at 100 days post-transplant is a critical point followed by transplant centers to assess the quality of their transplant program. Similarly, transplant centers keep records of the causes of deaths of the patients transplanted at their centers. Deaths that are not caused by relapse or progression of disease are said to be "transplant-related." Transplant-related deaths (mortality) within the first 100 days after a transplant are presumed to be due to the toxicities of the recipient's various pre-transplant therapies and/or the pre-transplant conditioning (chemotherapy, radiation) regimen.
The Advisory Board Company published 100 Day Transplant-Related Mortality benchmarks from the 2004 Oncology Roundtable of 5% for autologous transplants, 20% for allogeneic (related donor) transplants, and 40% for allogeneic (unrelated donor) transplants. The percentage of transplant-related mortality is a standard that transplant centers aim not to exceed. For example, if a transplant center performed 60 autologous transplants in a given year, their goal would be to have fewer than 3 deaths within the first 100 days post-transplant that were due to transplant-related toxicities and not due to relapse or progression of disease.
Wilmot Cancer Institute's Blood & Marrow Transplant Program meets the goals established by these benchmarks.